POSTDOC RESEARCH PROJECTS
1. Costs of resistance to an infectious parasite decrease with host age
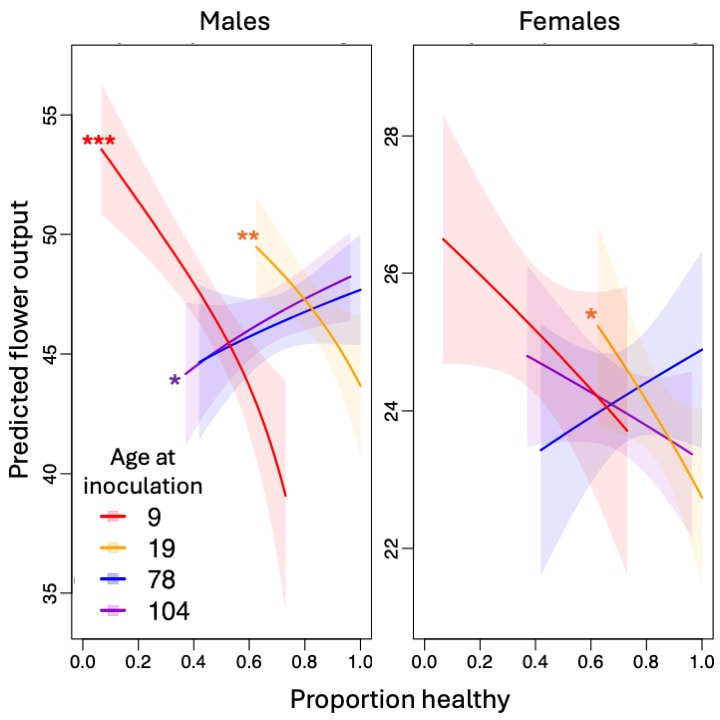
Many plants and animals are more susceptible as juveniles than as adults to infection by parasites. This pattern poses a puzzle for evolutionary biologists because hosts that get infected by virulent parasites as juveniles can potentially lose their entire lifetime fitness, hence selection should be expected to favor high resistance in juveniles. We tested the hypothesis that juvenile susceptibility is maintained by high costs of resistance at the juvenile life stage, and that juvenile resistance may be more costly than adult resistance. To test how the costs of resistance change with host age, we measured host susceptibility (proportion infected following a controlled exposure in the greenhouse) to the anther-smut fungus Microbotryum lychnis-dioicae at two seedling ages (9 and 19 days) and two adult ages (78 and 104 days) for 45 different full-sibling families of the host plant Silene latifolia. We also measured life history phenotypes of the same 45 host families in a seminatural field setting in the absence of disease for two years. We found significant variation among families in resistance at all ages. Susceptibility was highest at the seedling stage and there was no correlation between seedling and adult resistance. Resistance to infection was costly at two seedling stages, with high seedling resistance associated with lower flower production in the field. On the other hand, there was no association between adult resistance and flower production, suggesting that adult resistance was not costly. Our results support the hypotheses that resistance is costly, that resistance costs decrease with host age, and that costs of resistance could help explain the maintenance of juvenile susceptibility.